Research Planning
The Coronavirus Task Force research team plans to collect 600 blood samples from Japanese COVID-19 patients from our joint research institutes (95 hospitals as of June 15). Using these samples, we will conduct comprehensive analyses such as high-resolution HLA analysis (*6), SNP array analysis (*7), whole-genome sequence analysis (*8), and T-cell repertoire analysis. By comparing severe cases with mild and asymptomatic cases, we aim to identify the genes involved in the exacerbation of COVID-19 in Japanese patients.
Determining the target epitope for a vaccine is a critical step in vaccine development, but epitope prediction has proved to be a challenge. After identifying the genes involved in the exacerbation of COVID-19 in Japanese patients, the Coronavirus Task Force research team will aim to identify candidate antigens for SARS-CoV-2 using supercomputer simulations and will develop a vaccine better suited for preventing the exacerbation of the disease.
A series of genomic analyses have already been approved by ethics committees at Keio University and more than 30 medical facilities, and we have begun sequential sampling from authorized medical institutions while continuing to reach out to more medical institutions to cooperate.
Using proprietary molecular needle technology, the Coronavirus Task Force also aims to develop an effective mucosal vaccine (*9) for SARS-CoV-2. This vaccine will be based on information gained from genetic and immunological analyses of severe and mild or asymptomatic COVID-19 cases.
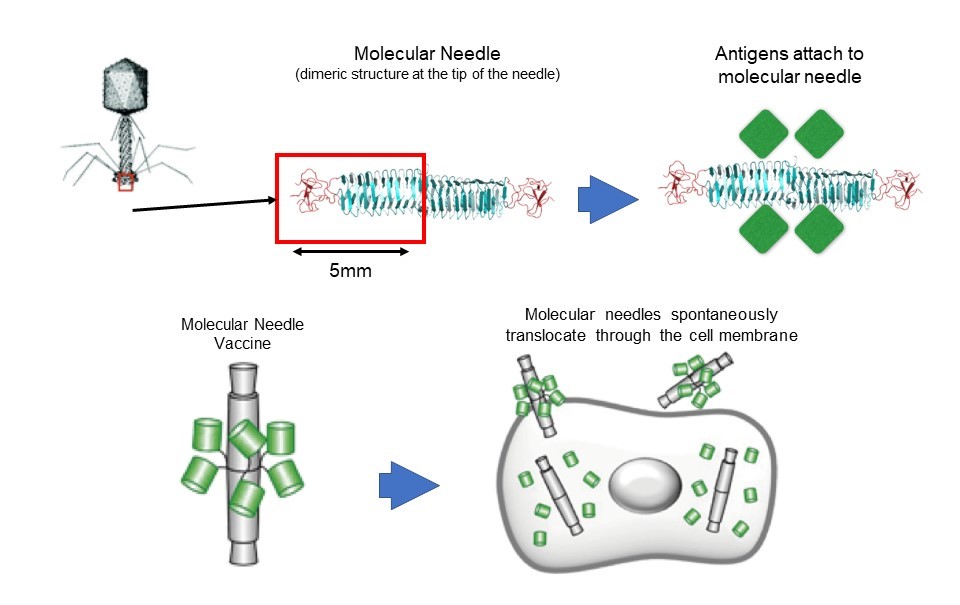
The molecular needle is a technology that transports protein molecules using a protein needle (molecular needle) approximately 10 nanometers in length at the end of the tail of a bacteriophage. Molecular needles can spontaneously translocate through a cell membrane and enter a cell, which makes it possible to deliver viral antigens to the body (inside of cells) to efficiently induce an immune response to a virus. To do this, we will take the proteins and peptide sequences of the viruses that comprise the vaccine and fuse them to the needle. Inoculations will then be administered intranasally via the mucous membranes of the nasal cavity or sublingually. Though conventional vaccines are mostly administered through subcutaneous injection, molecular needle technology can be administered by being inhaled through the nose, dripped under the tongue, or swallowed as a capsule, which means it is relatively safe and painless.
In this study, we will search for the genes involved in the exacerbation of COVID-19, but will also comprehensively examine the genes associated with COVID-19 immune responses. It is expected that the results will be used to effectively find a portion of the protein sequence of SARS-CoV-2—the virus that causes COVID-19—and will effectively prevent exacerbation of COVID-19 and help inform prevention and healing. We plan to develop a molecular needle vaccine that fuses the SARS-CoV-2 proteins and peptide sequences discovered in this way.